Analyses of the Ash Deposits
Rock Shelter Use and the Formation of the Ash Deposits; How did the Ash Deposits Form?
Several blocks of ash have been identified at this site. How the ash layers dating to the Initial Jomon period deposited in the rock shelter and the slope leading from the terrace were formed was estimated by analysing materials using three methods (1) thin section observation using a polarising microscope and X-ray diffraction, (2) X-ray fluorescence analysis, and (3) isotope analysis.
| 岩陰部灰層 | 第Ⅱ層群 |
①試料No.2・3(2・3は同じ灰層試料を分割;以下同じ) |
|
| 第Ⅲ層群 |
最上部 |
②試料No.4・5 (年代;9250〜9050 cal BP) |
|
|
上部 |
③試料No.6・7 |
||
| 前庭部灰層 | 10層 | 10a層 |
④試料No.13・14(年代;10200〜9500 cal BP) |
| 10b層 |
⑤試料No.15・16(年代;10200〜9500 cal BP) |
||
| 10c層 |
⑥試料No.17・18(年代;10200〜9500 cal BP) |
The first method involved observation of thin sections and X-ray diffraction. All of the ash deposit samples were embedded in resin and thin sections were cut and observed under a polarising microscope. The ash layer samples ② and ③ from inside the rock shelter were greyish-white in colour and whiter than the other samples, and analysis revealed that they were rich in calcite (crystals of calcium carbonate CaCO3), highly similar to the control sample, which was peach wood ash. Wood ash is mainly calcite, mixed with residues of plant tissue, charcoal, and surrounding geological deposits. Analysis of ash deposit sample ① from the inside the rock shelter and samples ④ to ⑥ from the terrace showed that they too were rich in calcite, but that they also contained plagioclase, pyroxene, andesite, rhyolite, altered volcanic rocks, and occasionally bone fragments, shells, and charcoal, suggesting that incorporations from the surrounding geology were present. X-ray diffraction showed large peaks of calcite in all samples, while in ash deposit samples ② and ③, other peaks were almost absent. On the other hand, in ash deposit samples ④ to ⑥ from the terrace area, there were plagioclase peaks as well as calcite peaks. This is consistent with the results of thin section observation.
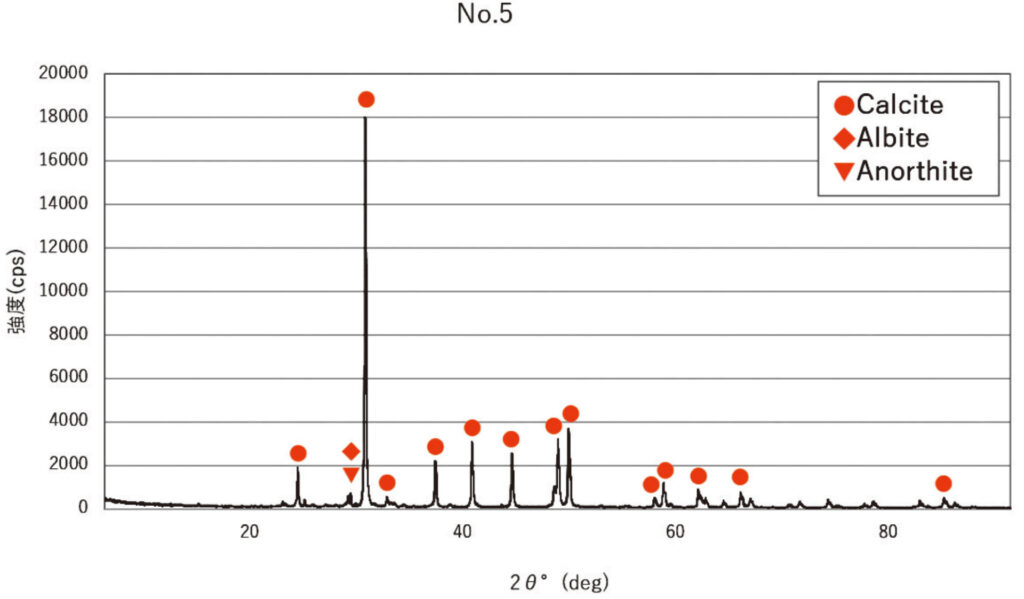
Calcite: large peaks of calcite crystals of calcium carbonate (CaCO3) were observed.
The second method employed was X-ray fluorescence analysis. Analysis of the types and amounts of the constituent elements showed that calcium (Ca) was abundant, especially in ash deposit samples ② and ③, which were found deep inside the rock shelter, with a content of around 85%. Potassium (K) is abundant in wood ash used for fertilisers, but here the concentration is 1-2%. The K content varies according to the type of wood burned and may have leached out into rainwater or groundwater. Ash deposit samples from the terrace area were high in silicon (Si) and aluminium (Al), suggesting that they may have been contaminated by the surrounding soil and other materials.
The third method employed was isotopic analysis. The carbon content of ash deposit samples ② and ③ was close to 10%. Pure CaCO3 contains 12% carbon, so it can be assumed they were mostly composed of calcite. The other ash deposits showed a carbon content of around 5%. The carbon isotope ratios δ13C, which indicate the proportion of carbon-13 (13C) present in the samples, were all around -25‰, which is consistent with the δ13C of C3 plants to which the majority of grasses and trees belong. The values are also similar to those of charcoal found together in the ash deposits.
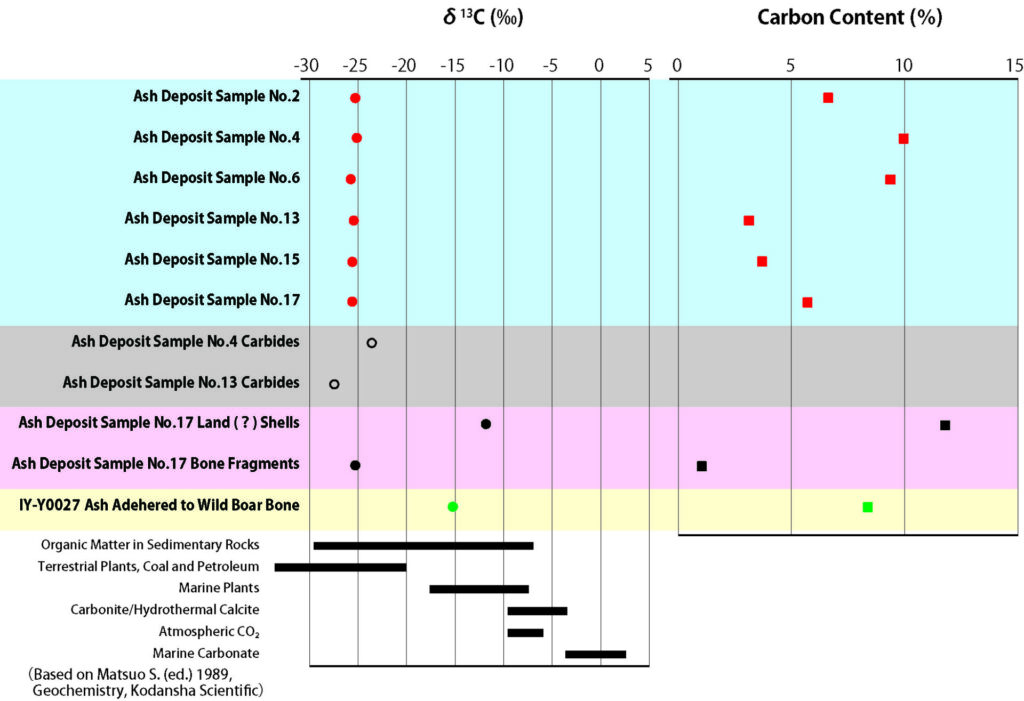
Explanation: Isotopes are atoms that have the same chemical properties as carbon and nitrogen,
but different physical properties in terms of mass. By comparing these proportions,
it is possible to estimate the origin of the substance and the way that chemical reactions proceed.
Summarizing the results obtained by the three methods, the formation of the ash deposits can be considered as follows.
It is known that calcite is the main component of the wood ash produced in bonfires.
Ca + O → CaO; calcium oxide
CaO + CO2 → CaCO3; calcite is formed by reaction with carbon dioxide
From the results of the analyses, it can be concluded that the ash deposits are derived from wood ash, rather than from field burning of C4 plants such as Kaya (Japanese nutmeg-yew) or from shells. In addition, as ash deposit samples ② and ③ are from inside the rock shelter, it is unlikely that they derive from natural forest fires.
Kunio Yoshida
Nobuo Miyauchi
Maiko Miura
Kasai Manabu
Shuji Ninomiya

